The Only Unified Solution for Your Oncology Service Line
Replace fragmented tools with the industry’s only end-to-end registry workflow. Eliminate backlogs, reduce staff burnout, and transform your registry into a strategic data asset.

Unlock the True Value of Your Oncology Data
Oncology service lines face an operational crisis, not just a technology gap. Rising case volumes and staffing shortages have turned the registry into a manual, high-cost burden. We solve this at the source.
Accurate, complete, and timely cancer reporting for state and federal compliance
Access idle patient data in a structured format to fuel research and discovery
Move beyond compliance to identify growth opportunities and drive patient retention.
Enhance operational efficiency to optimize staff workload, improve care delivery, and retain patients

The Only End-to-End Cancer Data Platform — Powered by AI for Hybrid Environments
Stop managing vendor handoffs. Our exclusive partnership with ONCO provides a single, automated workflow from the first clinical report to final submission.
By combining Inspirata’s advanced AI-driven technology with ONCO’s leading registry software and services, this collaboration streamlines the entire cancer data management process — from case finding to abstraction to reporting. The result is a unified, automated solution that empowers cancer programs to improve accuracy, efficiency, and outcomes across the board.
Efficient and Accurate
Casefinding

Automated


Streamlined

Seamless workflows
support


ONCOLOGY
AND REPORTING

to treatment
Efficient and Accurate
Casefinding
Automated

Streamlined

Seamless workflows
support

to treatment
Our Products
Who We Serve
Academic Medical Centers
Comprehensive Cancer Centers
NCI-Designated Cancer Centers
Oncology Departments
Community Oncology Practices
Lorem Ipsum Dolor



The Inspirata Difference

Accelerated Insight
Our proprietary technology autonomously mines clinical documents and identifies reportable cancer in real time with 98-99% accuracy*.
*findings based upon our standard commercial E-Path products and process
Built-In Best Practices
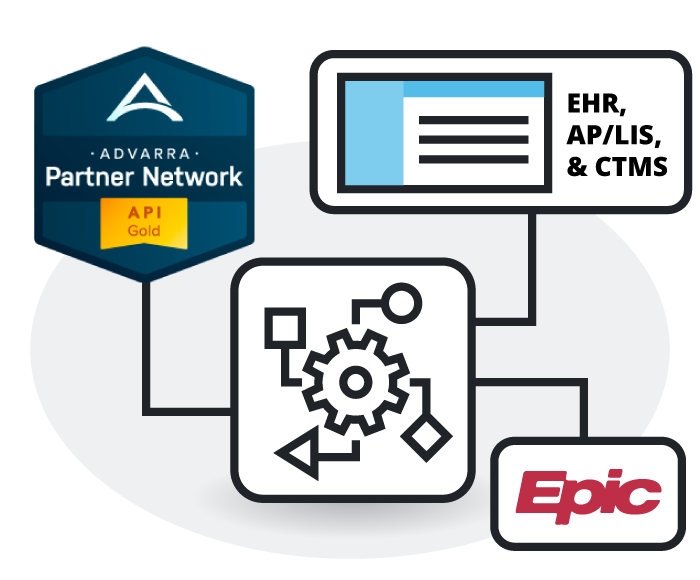
Interoperability
100% Oncology Focused

Frequently Asked Questions
How does an oncology data platform improve hospital operations?
A unified oncology data platform eliminates the silos between clinical systems and the registry. By automating the flow of data from the source (pathology and radiology) to the final report, hospitals reduce manual re-entry, minimize the risk of missed reportable cases, and allow oncology data specialists to focus on high-level quality assurance rather than administrative tasks.
What is the role of AI in cancer registry management?
AI and Natural Language Processing (NLP) are used to scan unstructured clinical documents such as pathology reports and imaging to identify reportable cancer cases instantly. This technology replaces manual casefinding, which typically takes 30-45 minutes per report, reducing it to seconds while maintaining 99% validated accuracy*.
*findings based upon our standard commercial E-Path products and process.
Can automated registry solutions help with oncology staffing shortages?
Yes. Automated solutions address the national shortage of oncology data specialists (ODS) by removing the most repetitive aspects of the registry workflow. Automation allows existing staff to manage higher case volumes without burnout, effectively scaling the registry’s capacity without requiring additional headcount.
Does the platform integrate with existing EHR and LIS systems?
The system is designed for broad interoperability, interfacing with major electronic health records (EHR), laboratory information systems (LIS), and radiology information systems (RIS). This ensures that data flows securely and directly from the clinical source into the registry workflow without manual intervention.
What are the primary benefits of an end-to-end cancer registry workflow?
An end-to-end workflow provides a single path from data capture to final stage and federal submission. This eliminates system handoffs, reduces the hidden costs of fragmented tools, and ensures that the data used for compliance is also available in real-time for clinical research and strategic planning.