Elevate the Efficiency and Precision of Your Oncology Operations
FASTER, EASIER, AND MORE ACCURATE ANALYSIS OF CANCER DATA

Trusted by Leading Healthcare Organizations
Leading Hospitals Trust Inspirata
Pathology, Radiology, and Related Clinical Reports Processed Annually
Customer Retention Rate
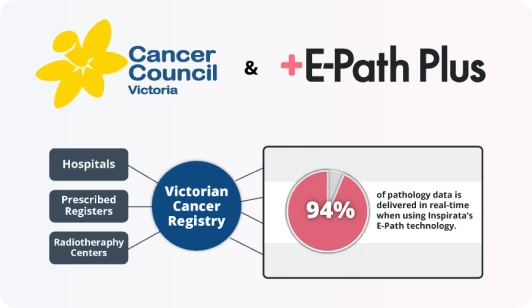
Unlock Your Cancer Registry's Full Potential
Empower Cancer Registry Staff
E-Path Plus facilitates accurate, complete, and timely reporting to augment the work of your cancer registrars and oncology data specialists. This helps reduce backlogs, create more efficient workflows, improve staff retention, and free up time for staff to focus on other high-impact activities for the facility.
Increase Operational Efficiency
Our AI/NLP engine utilizes advanced algorithms to accurately extract and analyze insights from extensive medical documentation effortlessly. Quickly identify significant trends, monitor essential KPIs, and gain insights to support clinical decisions, strategic planning, care quality, and research initiatives.
Optimize Costs.
Maximize Quality.
From mitigating staffing challenges to ensuring compliance to ever-changing data standards, E-Path Solutions help your cancer registry optimize costs while maximizing quality. Meet crucial reporting deadlines to prevent penalties and preserve your accreditation status with confidence.
Use Cases
Cancer Casefinding
Accelerate casefinding, minimize human error, and reduce manual workload. E-Path stands out in the market by identifying reportable cancer with an unparalleled 98-99% accuracy rate.
Intelligent Data Abstraction
Improve efficiency and accuracy to meet critical reporting deadlines. E-Path Plus can swiftly read, ingest, extract, and perform intelligent data abstraction for +190 data elements in mere seconds that can be used to demonstrate concordance to best practices.
Research and Discovery
Unlock cancer data’s full potential with E-Path Plus custom reports to effortlessly extract meaningful information, identify trends, and gain actionable insights to empower research, expedite publications, and bolster population health initiatives with data-driven confidence.

Revealed: The True Cost of Inefficient Cancer Registries
How It Works
Casefinding and Intelligent Data Abstraction
- Automated Document Analysis
- Real-time analysis of unstructured source documents
- Automated identification of reportable cancers
- Data Extraction
- Automated extraction and coding of applicable data elements
- Automated linkage of related documents for a single case
- Efficient Case Management
- Streamlined transfer of reportable cancer cases to registry(ies)
- Interactive portal for intelligent data abstraction, including tools for visual review and conflict resolution
- Direct in-application access to NAACCR Data Dictionary and code tables

Compliance, Quality, and Performance
- NAACCR-standardized and custom data collection forms and output formats
- Adherence to government and American College of Surgeon (CoC, NAPBC, NAPRC) reporting requirements
- Computer-assisted data extraction and monitoring for quality measures or performance improvement initiatives
Optimize Patient Retention and Outcomes
- Real-time identification of cases for multi-disciplinary cancer conference (tumor boards)
- Real-time identification of cases for nurse navigation and high-risk groups